|
Current research continues into the structure of the atom. These subatomic particles cannot be freed and studied in isolation. The more electronegative an atom, the tighter it pulls the electrons. An electron, like a proton is a charged particle, although opposite in sign, but unlike a proton, an electron has negligible atomic mass. With a lack of electrical charge, the neutron is not repelled by the cloud of electrons or by the nucleus, making it a useful tool for probing the structure of the atom.Įven the individual protons and neutrons have internal structure, called quarks. Electron: A negatively charged particle found circling or orbiting an atomic nucleus. The neutron carries no electrical charge and has the same mass as the proton. Electron is the negatively charged particle that rotating around the nucleus at the constant speed. It was discovered by a British physicist, Sir James Chadwick. The neutron is the other type of particle found in the nucleus. Which particle has a negative charge Easily put, an electron is a subatomic particle that carries a negative electric charge. A proton has a mass of 1.67 x 10 -27 kilograms. The number of protons in the nucleus of an atom determines what kind of chemical element it is. The proton's charge is equal but opposite to the negative charge of the electron. The existence of a positively charged particle, a proton, in the nucleus was proved by Sir Ernest Rutherford in 1919. One of the two types of particles found in the nucleus is the proton. The mass of an electron is 9.1 x 10 -31 kilograms. a beta particle In the reaction AI + HeP+ X, the particle represented by X is 4. Most properties of atoms are based on the number and arrangement of their electrons. Transcribed Image Text: CF4yTjq57vBxxhRY5wLzVymZP/view 3. The negatively charged electrons follow a random pattern within defined energy shells around the nucleus. Thomson discovered the existence of the electron, marking the beginning of modern atomic physics. The charge of a fundamental particle may be positive or negative, but its magnitude is always an integer multiple of the fundamental quantity qe 1.610-19. The diameter of an atom is approximately 2-3 Å. A convenient unit of length for measuring atomic sizes is the angstrom (Å), which is defined as 1 x 10 -10 meters. This nucleus contains most of the atom's mass and is composed of protons and neutrons (except for common hydrogen which has only one proton). 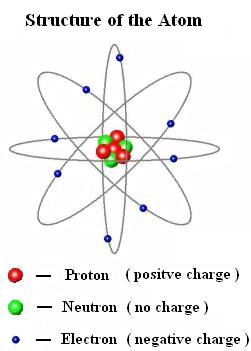
An atom is a complex arrangement of negatively charged electrons arranged in defined shells about a positively charged nucleus.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
